2020 News Archive
Second Review of my Economic Geology 2nd ed. Book Printed (Dec 12, 2020)
Economic Geology 2nd ed. (2020) Review by Professor Emeritus János Földessy of Miscolc University (Hungary), published in the Journal European Geologist 50, p. 90, November 2020.
Limited to one page, Professor Földessy reports on structure (7 Chapters) and contents of my book, of course briefly. He writes, for example: "Chapter 5 highlights the application of economic geology in dense 50 pages. Following the life of mine concept, topics range from the principles and methods of exploration such as the mineral systems concept, mineral prospectivity modelling, remote sensing, geochemistry, geophysics and drilling to resource estimation, the mining environment, mine closure, and the restitution of ecosystem services."
Földessy also notes that "Naturally in a book that condenses whole libraries, the depth of treatment is limited. For an advanced level of enquiry, abundant references to important printed and online sources are provided throughout. The referenced bibliography and Indexes are valuable components of the book. References occupy some 80 pages, offering a good start for deeper research. The General Index is lean with 14 pages but a detailed online version is in preparation. The Location Index takes us through the mentioned sites and mines within 12 pages. With some 300 Figures, the book is well illustrated and equipped with 81 Equations, 32 Tables, 25 Boxes, and 32 Colour Plates. Boxes feature case histories – relevant to important topics of the host chapter and illuminating text with maps and sections of deposits. Most equations illustrate chemical processes concerning ore formation."
In my view, in spite of the brevity, Professor Földessy provides highly useful information for evaluating the book. Thank you for your fine work, János!
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
Back to top
Home
First Review of my Economic Geology book 2nd ed. appeared in Print (Nov 27, 2020)
This review for the Journal Mine Water and the Environment (Springer) was written by Prof. Christian Wolkersdorfer. Christian is the founder of the International Mine Water Association (IMWA, see below).
I had hoped to learn from him, being a mine water specialist, how far my efforts to adress issues of the mining environment and mine water throughout the Economic Geology book, for example, in all metal sections are correct and valuable, such as in the introduction to 2.1.3 Chromium on page 273:
"Both for humans and for animals, chromium is an essential trace element (Lindh 2005). In higher concentrations, however, it is toxic and carcinogenic, especially in the hexavalent ionic state. Because chromic acid and its salts were widely used by small industry such as galvanization and tannery, chromium is a common groundwater pollutant. Another frequent source of pollution is chrome-plated waste in refuse dumps. Cr+6 polluted waters can be cleaned by passage through reducing, permeable barriers that are installed in the aquifer. Isotope analysis assists efficiency controls, because light Cr-isotopes are preferentially reduced (Blowes 2002, Ellis et al. 2002). Reduction leads to precipitation of insoluble Cr(III) hydroxide Cr(OH)3 and adsorption on oxy-hydroxides of iron and manganese."
Or if Section 5.4.2 Waste rock, tailings and seepage water: Acid Mine Drainage (AMD), starting on page 487, is correct and would be useful for readers of the Mine Water Journal. Unfortunately, this kind of information essential for the user of a Book Review is not available.
Wolkersdorfer concludes with kind words: Summarizing, I want to recommend this easy-to-read and valuable book to everybody in the world who is involved in economic geology and mining: students, teachers, researchers, professionals, NGO's, and administrators alike. Let me finish this review with Walter Pohl's words, which make clear why reading this book is important:
Well-managed extraction of minerals has every potential to contribute to the well-being of mankind, to increase material prosperity, to maintain a sustainable and vital social and natural environment, and peace.
Wolkersdorfer, Ch. (2020) INSIGHTS: Pohl's Economic Geology 2nd Edition (Book Review). Mine Water and the Environment https://doi.org/10.1007/s10230-020-00738-yIMWA or https://rdcu.be/ca92q
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
Back to top
Home
Mine Water Solutions -- Proceedings of the International Mine Water Association 14th Congress in New Zealand 2020 (November 20, 2020)
Read the message of the organizers: Welcome to IMWA 2020 New Zealand!, that is how we intended to greet you in Christchurch, the largest city on the South Island of New Zealand today. Sadly, we had to inform you that IMWA 2020 in New Zealand had to be postponed in response to disruption by the COVID-19 pandemic on global travel and economic conditions. We hope that the delay, though frustrating, only builds your anticipation and desire to visit New Zealand and participate in IMWA 2022 in Christchurch.
All of us were looking forward to a fantastic IMWA Congress in New Zealand this year. Though we can not hold said Conference, we don't want to leave you empty handed. Therefore we still compiled proceedings for the 14th IMWA Congress. 223 abstracts were accepted for presentation, and 40 of them are now published in these proceedings. They cover the full range of mine water related topics by experts from all around the world. Though you cannot listen to the presentations, we hope that you still can enjoy reading the related papers. They can be found at the link below.
Dear Reader, I, Walter, would ask you to note the title of the book: Mine Water Solutions. Understand this as a call for solutions, not only for uncovering mine water issues. And indeed, this book reports ongoing research and innovative methods to mitigate problems! For an easy search the book contains both an Author Index (p 243-244) and a Keyword Index (245-246). If mine water should be one of your frequent occupations, consider joining the International Mine Water Association and regularly reading their journal Mine Water and the Environment (MWEN). My new Economic Geology (2020) book also is a prolific source of information concerning mine water, and mitigation of water and environmental risks, in the context of ore deposits..
Pope, J., Wolkersdorfer, Ch., Sartz, L., Weber, A., Wolkersdorfer, K. (2020) Mine water solutions. 268 pp. International Mine Water Association 14th Congress New Zealand 2020 Proceedings. ISBN 978-3-00-067297-2 https://www.imwa.info/docs/imwa_2020/IMWA_2020_proceedings.pdf OPEN ACCESS..

- Rio Tinto (the red river), Southern Spain, some kilometers downstream from the former mining area where AMD and ARD make up most of the inflow. Today, the old mine buildings, an ancient railway and the brownfield landscape is a popular touristic park (https://parquemineroderiotinto.es/). For professionals, it is an example of the evolution many decades after the mine was abandoned. Modern Green Mining would, of course, remediate the area, although the Rio Tinto mines might hold resources for a revival that could pay for reclamation.
Back to top
Home
Adakites and Copper-Gold-Tellurium Deposits — Exploration Key Throughout Earth History? (September 25, 2020)
When writing my Economic Geology (EG2) book, I noticed an increasing number of reports of adakites and adakitic magmatic rocks at ancient gold deposits. A striking example is the giant Archean Golden Mile Au-Te deposit, Kalgoorlie, Western Australia, where Mueller et al. (2016) found 'weighty arguments that support a significant magmatic component of adakitic affinity in the hydrothermal system at Kalgoorlie' (Pohl 2020, Box 2.4, pages 234-235).
Adakites have first been recognized (and named) at Adak Island, Aleutian Islands (Defant & Drummond 1990). Very soon, their close genetic relation with intrusion-related gold-telluride deposits prevalent in Cenozoic magmatic arcs made the presence of these rocks one of the exploration guides for porphyry Cu-Au and epithermal deposits.
Adakites are geochemically distinct andesites to dacites. At Adak, they are characterized by highly fractionated REE, high La/Yb, high Sr and a lack of a Eu anomaly, indicating partial melting of subducted oceanic crust at ~ 50-100 km depth; the high pressure imposing garnet in the restite (Figure 1). Meanwhile, several variants of the tectonic setting of adakites have been proposed.

- Figure 1 Garnet cloud (idiomorphic, <8 mm) in ophiolitic metabasalt in a shear zone (nappe boundary) within crystalline basement of the Variscan chain in Central Europe may illustrate restite after adakite melt abstraction. Note coin (black dot) for scale (D = 2 cm).
Deng et al. (2020) returned to the volcanic arc exposed in the Philippines that hosts a number of important adakite-related Cu-Au deposits. Their paper provides an up-to-date description of the tectonic setting, the deposits, and of the age and geochemistry of adakites and country rocks. Using data published elsewhere, they employ current petrogenetic models such as slab melting, fractional crystallization of minerals such as clinopyroxene, hornblende, apatite, and titanite, and magma mixing for the ore-bearing adakitic rocks and coexisting basaltic rocks, in order to elucidate origin and evolution of adakites and their relation to the mineralisation in Northern Luzon.
The general tectonic setting of the Philippines is eastward subduction of the oceanic South China Sea Plate beneath the Philippine Arc (Figure 2). Note the Scarborough Seamounts at ca. 15o latitude; the greater size of the Cu-Au deposits in Northern Luzon is thought to be due to the subduction of this ridge. Northern Luzon contained >70 Moz of gold and several million tonnes (Mt) of copper in porphyry, epithermal, and skarn deposits; Deng et al. (2020) tabulate data of nine important deposits.
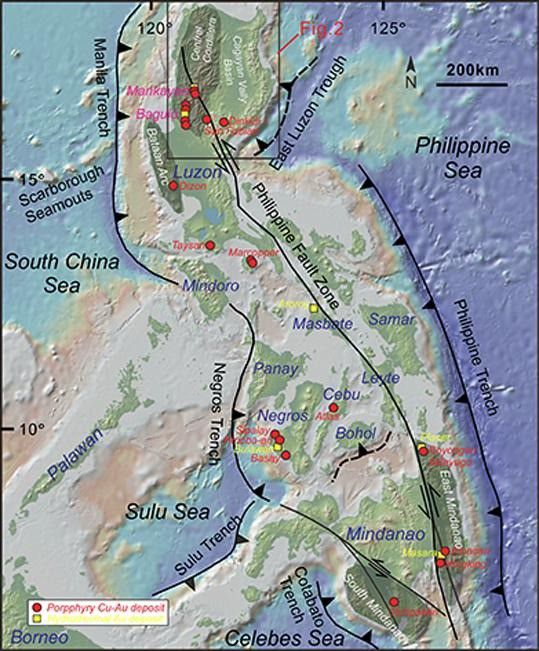
- Figure 2 Tectonic sketch map of the Philippine archipelago, with locations of porphyry Cu-Au (red solid circle) and epithermal Au (yellow solid square) deposits. Courtesy Deng et al. (2020). Base map is from GeoMapApp (http://www.geomapapp.org) and tectonic lines are from Aurelio and Pena (2010).
The origin by slab melting and the resulting high oxygen fugacity of adakites are the most critical factors for Cu-Au mineralization (Sun et al. 2012). Cu mineralization is favoured by adakites being partial melts from subducted slabs, given the high initial Cu tenors of oceanic crust at 70 ppm (fresh basalt: EarthRef.org), high sulfur and high oxygen fugacities of slab-derived melt. MORB contains 0.75 plus/minus 0.5 ppb Au (Pitcairn et al. 2015). Arsenian iron sulfides of mid-ocean ridges concentrate gold and silver (28-140 ppm Au, 800-2400 ppm Ag: Halbach et al. 2003).
Deng et al. (2020) conclude that two different tectonic settings, one distensive (a) and the other contractive (b), formed copper-gold deposits in Northern Luzon:
a) The Late Oligocene alkalic Dinkidi stock is a highly differentiated adakite evolved from basaltic magma, which was derived from partial melting of slab melt metasomatized mantle in a backarc rift setting. The enriched source, and later enrichment of metals by crystal fractionation produced the Dinkidi alkalic porphyry Cu-Au deposit.
b) Pliocene-Pleistocene dioritic to dacitic porphyries in the Central Cordillera show transitional to typical adakite signatures. Partial slab melting at a shallow depth was probably caused by Scarborough Ridge subduction beneath the Luzon arc giving rise to epithermal Lepanto and several sizeable porphyry Cu-Au deposits.
Exploration in modern and old volcanic arcs may profit through applying the understanding contributed by this paper.
References
Aurelio, M.A. & Pena, R.E. (2010) Geology of the Philippines. Mines and Geosciences Bureau, Quezon City, pp. 1-532.
Deng, J.H., Yang, X.Y., Zhang, L.P., Duan, L.A., Mastoi, A.S., Liu, H. (2020) An overview on the origin of adakites/adakitic rocks and related porphyry Cu-Au mineralization, Northern Luzon, Philippines. Ore Geol. Rev. 124, 22 pp. https://doi.org/10.1016/j.oregeorev.2020.103610.
Defant, M.J., Drummond, M.S. (1990) Derivation of some modern arc magmas by melting of young subducted lithosphere. Nature 347, 662-665.
Halbach, P.E., Fouquet, Y. & Herzig, P. (2003) Mineralization and compositional patterns in deep-sea hydrothermal systems. Pp 85-122 in Halbach, P.E., Tunnicliffe, V. & Hein, J.R. (eds) Energy and mass transfer in marine hydrothermal systems. Dahlem Workshop Report 89, 365 pp.
Pitcairn, I.K., Craw, D. & Teagle, D.A.H. (2015) Metabasalts as sources of metals in orogenic gold deposits. Miner. Deposita 50, 373-390.
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons — an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
Sun, W.D., Ling, M.X., Chung, S.L., Ding, X., Yang, X.Y., Liang, H.Y., Fan, W.M., Goldfarb, R., Yin, Q.Z. (2012) Geochemical Constraints on Adakites of Different Origins and Copper Mineralization. J. Geol. 120, 105-120.
Back to top
Home
NEW BOOK, NEW ENTRY IN MY ECONOMIC GEOLOGY BLOG (August 15, 2020)
JUST APPEARED
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons —
an Introduction to Formation and Sustainable Exploitation of Mineral Deposits.
2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
This Economic Geology book evolved from an earlier edition (EG1) that appeared in 2011. Its structure is similar to the precursor, but the contents have been profoundly updated. The new edition of the book (EG2) reflects the great advancement of science and practice in the years since.
With 32 Colour Plates, 305 Figures, 32 Tables, 25 Boxes, and 81 Equations, the new edition is only a little more voluminous than EG1. An abundance of new and a reduced number of older references fill nearly 80 pages. The List of Boxes appears on page 727. The printed General Index spreads across 16 pages; the author intends to make more entries in the future and to provide the extended version on his website. The Location Index is a new feature of the book that fills 12 pages of small print. Apart from numerous long-known type deposits such as Kiruna in Sweden or the Golden Mile in Australia, the reader may expect many new entries.
For students and teachers, there will soon be a companion page with supporting matter on the website of the publishers. The Table of Contents, a flyer and Sample Pages can be accessed at URLs
www.schweizerbart.de/9783510654352 (Soft Cover)
www.schweizerbart.de/9783510654413 (Hard Cover)
WALTER L. POHL is Emeritus Professor, a science writer and consulting geologist in economic geology. For more information visit: htpps://www.walter-pohl.com
Back to top
Home
Hypogene Hydrothermal Ultramafic-Hosted Magnesite and Engineered Carbon Dioxide Removal (CDR) from Air by Fixation in Ultramafic Rocks: a Broad Investigation. OPEN ACCESS (July 9, 2020)
Are you interested to learn more about one of several geological methods of climate intervention? Or about magnesite? - – A technical review-type paper by Kelemen et al. (2020) provides useful information and proposes urgent lines of research.
The authors point out that the Intergovernmental Panel on Climate Change (IPCC) foresees that soon, annual removal of more than ten gigatons (Gt) CO2 from air will be required to keep global warming below 1.5 to 2 degrees C (IPCC 2018). This report introduces the term 'Carbon Dioxide Removal' (CDR). If you should not yet have read any material from IPCC, this may serve your initiation.
Reactive rocks are ultramafics rich in Mg and Ca, in minerals such as olivine, pyroxene, serpentine and brucite. Useful reactions are of the type
Magnesite formation from dunite (Pohl 2020) (eq.1)
Olivine (forsterite) Mg2SiO4 + 2 carbon dioxide CO2aq → 2 Magnesite MgCO3 + Silica SiO2aq
This reaction is one of the potential pathways to mineralize carbon from CO2 in air. It is spontaneous, relatively rapid and exothermic. The energy density of unreacted olivine in the presence of H2O or CO2 at low temperature is ~2 GJ per cubic m, only ~20 times smaller than the energy density of gasoline (Kelemen et al. 2020). This naturally available reservoir of chemical potential energy can be used to reduce external energy inputs and costs for CDR.
The CO2 uptake and storage capacity for complete carbonation in these materials is significant: 0.62 tonnes of CO2 per tonne of olivine reactant, 0.76 t of CO2 per tonne of brucite reactant, and ~0.4 to 0.5 tonnes of CO2 per tonne of pyroxene, serpentine, wollastonite, and industrial waste similar to wollastonite in composition. The resulting magnesite and other carbonates is nearly insoluble; sequestering CO2 for geological times.
Kelemen et al. (2020) inform on details such as the cost of grinding the rocks, direct use of mine tailings (from operations producing Cr, Pt, Ni, Cu, diamonds etc.), and using magnesite for 'MgO + CO2 cycling or looping', which would include geological sequestering of carbon dioxide. In situ circulating of aqueous CO2 solutions in ultramafics and basalt (projects in Iceland and Washington State) may become significant. The CarbFix project in Iceland is given room for a more detailed description; it safely sequesters all CO2 from local thermal power stations in basalt.
The natural paradigm is the magnesite deposits of the 'Kraubath' type that are principally the product of the reaction between ultramafics and CO2-rich aqueous fluids (eq. 1) (Pohl 2020). Weathering, for example, results in hyperalkaline groundwater that may dissolve atmospheric CO2 resulting in rapid precipitation of carbonate minerals such as magnesite and hydromagnesite (Pohl 2020). Nearly all outcrops of ultramafic rocks display carbonate crusts and thin joint fills, which illustrate the ease of carbonatization in contact with the atmosphere. In fact, this observation inspired the proposals to employ reaction eq. 1 for carbon dioxide sequestration.

- Dense white 'bone' magnesite with a little quartz (grey, translucent) filling fractures in dunite is a product of near-surface meteoric or low-temperature hypogene carbon dioxide waters reacting with olivine (Kinyiki Hill, Tsavo, Kenya).
The prime agent of atmospheric CO2 drawdown appears to be the obduction of ophiolites, combined with tropical weathering, and consequent carbonatisation of ultramafic and mafic rocks, as demonstrated by Macdonald et al. (2019) for the last 500 My, repeatedly triggering icehouse Earth conditions. In this connex you may be interested in the latest edition of the CO2 diagram in the atmosphere since 400 Ma (Foster et al. 2017). Find more detail in my blog 'Mining, Ophiolites, CO2 and Climate Change: an Astonishing Discovery' (January 10, 2019).
Kelemen et al. (2020) briefly describe geological examples of natural carbon mineralisation in the Semail ophiolite (Oman), marked by large travertine deposits and listvenites (also written listwaenites; this term describes hydrothermally altered carbonated and silicified ultramafic rocks that may be associated with epigenetic gold mineralization; the altered rocks consist of magnesium-calcium-iron carbonates, chromian muscovite and quartz, with various accessory minerals: Pohl 2020).
Kelemen et al. (2020) touch on the problem that in situ carbonatization may lead either to reaction-driven cracking or to clogging of flow paths; this section should be of great interest to investigations of ultramafic-hosted magnesite deposits. The role of alkaline water in the natural process system is explained. The authors write that
Olivine-rich peridotite could, in principle, consume >1 Gt CO2 per cubic km peridotite per year, at temperatures greater than ~150 degrees C, and CO2 partial pressures greater than ~60 bars
!Notice that this sentence may describe the typical formation conditions of hypogene hydrothermal ultramafic-hosted magnesite!
Inducing thermal convection should be employed in order to reduce costs. Drillhole-based projects depend on rock mass permeability. For ultramafic massivs, this is largely unknown and Kelemen et al. (2020) use model calculations. Geological observations in ultramafic-hosted magnesite mines might contribute to close this gap. Here, I found another remarkable statement:
The common result (is) that disposing of fossil fuel emissions requires infrastructure with a scale similar to that of fossil fuel extraction.
Kelemen et al. (2020) also consider spreading reactive material on land for CO2 removal from air; based on basalt, this theme is more broadly treated by Beerling et al. (2020). Unusual for science papers, Kelemen et al. (2020) provide a substantial section (6.) discussing possible research lines.
Apart from the valuable details that are provided in these papers, the general trend is of interest. In the recent past, interfering with climate by geoengineering was considered unethical. Now, geoengineering enters mainstream science. This is progress in the sense of the Enlightenment.
References
Beerling, D.J., Kantzas, E.P., Lomas, M.R. et al. (2020) Potential for large-scale CO2 removal via enhanced rock weathering with croplands. Nature 583, 242-248 (2020). https://doi.org/10.1038/s41586-020-2448-9
Foster, G.L., Royer, D.L. and Lunt, D.J. (2017) Future climate forcing potentially without precedent in the last 420 million years. Nature Communications 8, 14845. OPEN ACESS. DOI: 10.1038/ncomms14845
IPCC (2018) Global Warming of 1.5oC. Intergovernmental Panel on Climate Change. URL https://www.ipcc.ch/sr15/
Kelemen, P.B., McQueen, N., Wilcox, J., Renforth, Ph., Dippled, G. and Paukert Vankeuren, A. (2020) Engineered carbon mineralization in ultramafic rocks for CO2 removal from air: Review and new insights. Chemical Geology 550, 119628. OPEN ACCESS. DOI: https://doi.org/10.1016/j.chemgeo.2020.119628
Macdonald, F.A., Swanson-Hysell, N.L., Park,Y., et al. (2019) Arc-continent collisions in the tropics set Earth's climate state. Science 364, 181-184. DOI: 10.1126/science.aav5300
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers (the book is due to appear in ~one weeks time).
Back to top
Home
GREETING GEOETHICS (June 17, 2020)
The philosophical foundations of Geoethics, as propagated by the International Association for Promoting Geoethics (IAPG), are the principles of the Enlightenment: Reason, Science, Humanism and Progress. IAPG's website welds contemporary ethics (moral philosophy) with current geoscience and its practice. The latest broad treatment of the subject seems to be the book edited by M. Bohle (2019). Many of the professional societies such as the Geological Society of America have a short 'Code of Ethics' for members. I suggest that the essentials of Geoethics should be part of the Earth Science syllabus at universities.
As an Economic Geologist I believe that universal improvement of the human condition inevitably requires mineral raw materials. In my new book EG2, which is about to appear, my plea is for responsible, 'green mining'. Scientific advice towards that goal is provided throughout. Current best practice is illustrated by the great international mining giants. Sadly, some countries that rank among the lowest in political stability, regulatory quality, and control of corruption trail far behind.
Yet, globally, steady improvement is taking place although we must not relent in our efforts.

- Best practice remediation: Post-mining lignite open pit lakes in the Lausitz region, Germany, in the last stages of filling and rehabilitation. Note the wind power plants. Courtesy of P. Radke, © LMBV, Lausitzer und Mitteldeutsche Bergbau-Verwaltungsgesellschaft mbH.
Read the last sentence of the Epilogue to my EG2:
In conclusion, allow me to reiterate that well-managed extraction of minerals has every potential to contribute to the well-being of mankind, to increase material prosperity, to maintain a sustainable and vital social and natural environment, and peace.
WALTER L. POHL is Emeritus Professor, a science writer and a consultant in economic geology.
References
Bohle, M. (ed) (2019) Exploring Geoethics: Ethical Implications, Societal Contexts, and Professional Obligations of the Geosciences. 214 pp. Palgrave Pivot, ISBN 978-3-030-12009-2.
International Association for Promoting Geoethics (IAPG) URL http://www.geoethics.org/ Last accessed June 15, 2020.
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
Back to top
Home
A Compact Guide for Mine Water Management — Open Access (May 20, 2020)
Water issues in mining are central. Generally, a well-planned water management is indispensable throughout the whole life-cycle of every mine, from the stage of feasibility and environmental impact studies to post-closure monitoring. Mine-site water management should always consider the entire catchment. A worldwide investigation of past mine water studies presented as part of environmental impact statements revealed significant errors and often, underestimated impacts. Hydrological studies, for example, were often based on data of only one annual cycle. It is impossible, however, to predict extreme precipitation and flooding or drought from one year's data. Yet, extreme rainfall and snow-melt are the common cause of dam breaks, landslides and mudflows. Clearly, upper and lower bounds of all hydrologic conditions affecting a mine must be determined. Best practice approach is to establish a monitoring network that assembles climate, surface and groundwater data. Initial models of water quality and flow are continuously updated. — The above text is an excerpt from my new Economic Geology book (in short EG2), Section 5.4.1 Potential environmental problems and solutions related to mining and mine-site processing (Pohl in print 2020, p. 482 ff). Many more entries concerning mine water appear throughout the EG2.

- Famous contaminated Río Tinto river some kilometers downstream of the large brownfield Río Tinto VMS copper mining district in SW Spain. The photo dates from the wet season, when the water is not red and the pH tends towards neutral. Riparian vegetation is healthy but the banks are marked by ochreous iron oxy-hydroxides such as hydronium jarosite (yellow) and goethite. Such rivers can be remediated by limestone sand treatment. Río Tinto's unique, rich fluvial biodiversity of extremophiles may, however, deserve protection (Pohl 2020).
Today, I present a recent paper by Wolkersdorfer et al. (8 co-authors) (2020). Initially written as a report on guidelines for IAEA (International Atomic Energy Agency, Vienna) the paper aims to 'summarize international state-of-the-art applications and opportunities for employing and deploying hydrological, geochemical, and isotopic tools in an integrated manner for investigations of mining operations'. The guide is written for 'scientists (including isotope specialists) who have limited or no mine water experience, environmental managers, planners, consultants, and regulators with key interests in planned, active, and legacy mining projects'.
Content is organized into 'Introduction', 'How to Consult the Literature', a highly useful list of 'Mine Water Relevant Databases and Literature Repositories', the 'Natural Background', a section on 'Mine Water: Fundamentals of Mine Drainage Quality', which includes 14 chemical formulas that illustrate abiotic and biotic sulfide oxidation. The resulting waters are commonly acidic and carry dissolved sulfate, iron and other metals. pH and the composition of global examples of 16 such waters are provided in Table 1. Nine minerals that buffer the acidity are listed in Table 2. Frequent precipitates of mine waters are briefly described and the complex spatial-temporal variation of these systems is adressed. The section 'Mine Site Characterization' relates ore deposit classes such as magmatic-hydrothermal or VMS (volcanic massive sulfide) to the potential to develop AMD (acid mine drainage) or ARD (acid rock drainage), and metal pollution. A similar control exert different variants of hydrothermal alteration; advanced argillic alteration of porphyry Cu ore is a poor buffer of acidity, whereas propylitic gangue effectively controls it.
Further headings include 'Mine Site Hydrology', 'Components of the Water Balance', 'Site Conceptual Model Development Process', 'Strategic Approach', 'Environmental Impact Assessments', 'Components of a Mine-Site Water Perspective', 'Mine Tailings', 'Waste Rock Dumps', 'Heap Leach Materials', 'Overview of Mine Waste', 'Drainage from Mine Workings', 'Process Water', 'Water Treatment', 'Tracer Tests', 'Site Monitoring and Modelling', and 'Specific Isotopic Tools' (C, H, O, S, Pb, Hg, Cu, Ni, Mo, Zn).
Generally, the paper has the character of an overview but five pages of references support search for details as do brief case histories and some figures. I believe that even specialists in one of the adressed sectors will find the broad overview highly useful. I certainly do.
References
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons — an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
Wolkersdorfer, Ch., Nordstrom, D.K., Beckie, R.D., et al. (2020) Guidance for the Integrated Use of Hydrological, Geochemical, and Isotopic Tools in Mining Operations. Mine Water Environment, 25 pp. Open Access (Creative Commons Attribution 4.0), URL https://doi.org/10.1007/s10230-020-00666-x
Back to top
Home
A revolution, not only in gemstone science (April 18, 2020)
Could you guess what is common to gemstones (tsavorites) in E-Africa and to emeralds in the Eastern Alps in Europe? Answer: It is that they crystallized from a high T and P supercritical hydrous melt.
This is the revolution in gemstone science: Fluid inclusions (FIs) were investigated and described at length, but in many cases, swarms of melt inclusions (MIs) in the same samples remained unnoticed. These MIs, however, are the key to understanding the generation of gems. Could it be that teachers of fluid inclusion methods neglect this field?
Generally in published economic geology papers, melt inclusion research methods and data are rarely reported (Pohl 2020). Yet, in magma-related or high-grade metamorphic anatectic systems, melt inclusions (MI) preserve the chemical composition of the parental or partial melt, including volatiles and trace elements. This essential information is largely lost in fluids evolving from these melts. Investigating MIs comprises the determination of minerals and other phases, remelting and homogenization, quenching and analyzing the bulk melt-fluid system (Rainer Thomas in Hurai et al. 2015).
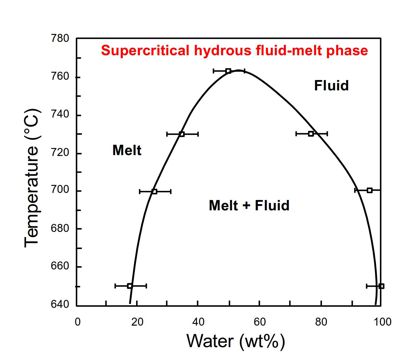
- Miscibility of water and a simple silicate melt (albite NaAlSi3O8) at 14.5 kbar. Modified from Shen & Keppler (1997). Note that in this system, there is complete miscibility above the solvus crest at 763 degrees C, in the field of a supercritical hydrous fluid-melt phase.
The key to formation of valuable gemstones is that above the critical T and P, water and liquid are completely miscible and merge into a supercritical hydrous fluid-melt (SHF-M) phase (Figure). Above the solvus crest supercritical water-rich liquids (the fluid/melt phase of Thomas et al. 2018 and Thomas et al. 2020), have properties that differ from subcritical state: Their density varies widely with changing pressure and temperature, they have higher pH, are able to dissolve many organic substances, and exhibit extreme dissociation of water, high diffusion rates and element fluxes, low dynamic viscosity and very low surface tension values. Geologically most important are probably the variable density (from dense fluid/melt to gas), reaction rates and equilibrium constants that control transport and precipitation (Thomas et al. 2020).

- Photo of plumasite-related barrel-shaped turbid rubies collected by Walter Pohl at John Saul's Mine, Mangari, SE Kenya.
Early turbid crystals grow from the supercritical hydrous melt (Photo Mangari Rubies) whereas transparent gems form from evolving fluids (Pohl 2019, 1985).
The significance of supercritical hydrous fluid/melt (SHF-M) phases in general metallogenesis is sadly underexplored. In recent models on supra-subduction formation of orogenic gold deposits, for example Groves et al. (2020), simple fluids are shown to effect the transport from a dehydrating slab into structural traps of the accreting orogen. Much more likely is the involvement of SHF-M phases!
References
Groves, D.L., Santosh, M., Deng, J., et al. (2020) A holistic model for the origin of orogenic gold deposits and its implications for exploration. Miner. Deposita 55, 275-292. https://doi.org/10.1007/s00126-019-00877-5
Hurai, V., Huraiova, M., Slobodnik, M. & Thomas, R. (2016) Geofluids - Developments in Microthermometry, Spectroscopy, Thermodynamics, and Stable Isotopes. 504 pp. Elsevier.
Pohl W.L. (2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. ~766 pp. Schweizerbart Science Publishers, Stuttgart.
Pohl, W.L. (2019) The tsavorite (gem quality vanadium grossular) minerogenetic system in East Africa. Arabian J. Geosciences 12, 612-618. https://doi.org/10.1007/s12517-019-4744-y
Pohl, W.L. (1985) Contributions to the origin of the Mangari gem corundum (ruby) deposits (SE Kenya). Pp 58-65 4 Figures in M.P. Tole (ed) Proceed. 1st Conf. Geol. Soc. Kenya (1984) GSK no. 2, Nairobi. (You can find this in my ResearchGate profile or ask me by email).
Thomas, R., et al. (2018) Genetic significance of the 867cm-1 out-of-plane Raman mode in graphite associated with V-bearing green grossular. Mineralogy & Petrology 112(5), 633-645. https://doi.org/10.1007/s00710-018-0563-1
Thomas, R., et al. (2020) Emerald from the Habachtal: new observations. Mineralogy &Petrology https://doi.org/10.1007/s00710-020-00700-4
Shen, A.H. & Keppler, H. (1997) Direct observation of complete miscibility in the albite-H2O system. Nature 385, 710-712.
Back to top
Home
Professorship of Metallogeny – Chair position at Laurentian University, Canada (March 21st, 2020)
Metallogeny? What is that? Is it not maps full of strange coloured symbols that are only good for hand waving when starting a presentation? -- Yes indeed, metallogenetic maps were part of the achievements of a past generation. Yet imagine the feat to assemble geology, locations, facts and data on mines and mineral deposits during the Cold War, working with and meeting professionals from both sides! My mentor, W.E. Petrascheck was one of those pioneers (see my photo gallery).
- Clipping of a Metallogenetic (often shortened to 'metallogenic') Map, here SW Europe (Juve & Storseth 1997). With kind permission by NGU/Trondheim.
At the time of the pioneers already, Metallogenetic Maps and supporting Mineral Database collections for all deposits were expected to assist both science and the search for new deposits. In retrospect, the promise was fulfilled by a remarkable discovery wave in the western realm (e.g. Chilean Cu giants, Olympic Dam, new mines in the Mt. Isa district, Voisey's Bay) and in China exploding soon after Mao's Cultural Revolution. The term 'metallogeny' (or metallogenesis) continues to be used, but often erroneously. It is not the genetic description of an individual deposit, but is understood as 'the science of the origin and distribution of ore deposits in geological space and time' (Pohl in print). This is the reason why it is so useful for mineral exploration.
Although metallogeny must have been one factor in the great mineral discovery period of the second half of the 20th Century, metallogenetic research began to fade away from the front pages of ore geology science and practice. Nobody had lauded its great contribution to understanding and to discoveries. A plethora of new technologies became available and producing data at all scales, from remote sensing to isotope geochemistry, took centre stage.
Initially hardly noticed, some three decades ago in Australia, a new approach to mineral exploration was developed emulating the well-proven 'petroleum system' concept of oil exploration (Wyborn et al. 1994). Called 'mineral systems', several variations of the method are currently the main tools for predictive prospectivity (or 'ore potential') mapping. Mineral system analysis requires a holistic genetic investigation that identifies all factors and processes, which contribute to the origin and location of mineral deposits. In practice, it evolved parallel with the digital revolution. For exploration, models of targeted deposit classes or types are based on geological knowledge, the underlying mineralization processes and their mappable features, data of known deposits, and probability mathematics. Systematic regional grid-sampled data include geology, geochemistry, geophysics (magnetic, gravity, electromagnetic, seismic and deep-sounding magnetotelluric tomographic surveys), and remote sensing. Data are processed in Geoscience Information Systems (GIS), either knowledge driven, or data driven with probability math methods such as fuzzy logic, nearest neighbour, weights of evidence and artificial neural networks. If you are looking for details, I recommend the paper by Skirrow et al. (2019), which describes an application of the mineral system method for IOCG mineral potential mapping in Australia. Note that access to this paper is OPEN.
You may also consult my blog October 15, 2019.
Digitization and the systematic approach propelled metallogeny into the present time (DIGITAL METALLOGENY), based on ever improving understanding of tectono-metallogenetic systems, of mineral deposit formation, and of skilful manipulation of big data. Output allows testing different genetic models (Skirrow et al. 2019), improving them, and delivering decision-support tools for delineating, ranking, and prioritizing exploration targets. Founded on digital quantitative metallogeny, shall we enter a new period of discovery successes?
The Metallogeny Chair at the Laurentian University is waiting for excellent candidates. The winner may expect over 100 million Can$ to work with. Does this tempt you? You may read all the details at the URL
Research Chair in Metallogeny, Laurentian University
First published in October 2019, the add re-appeared in the last edition of GSA Today (March-April, Geological Society of America). Applications are still sought. You may learn more from Dr. Doug Tinkham at dtinkham@laurentian.ca.
References
Juve, G. & Storseth, L.R. (eds) (1997) Europe and Neighbouring Countries. Sheet no. 9 of Mineral Atlas of the World at the scale of 1:10 000 000. Published by CGMW (Paris) and NGU (Trondheim).
Pohl W.L. (in print, 2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. 755 pp. Schweizerbart Science Publishers, Stuttgart.
Skirrow, R.G., Murr, J., Schofield, A., et al. (2019) Mapping iron oxide Cu-Au (IOCG) mineral potential in Australia using a knowledge-driven mineral systems-based approach. Ore Geology Reviews 113, 103011. Open access. https://doi.org/10.1016/j.oregeorev.2019.103011
Wyborn, L.A.I., Heinrich, C.A., Jaques, A.L. (1994) Australian Proterozoic mineral systems: essential ingredients and mappable criteria. The AusIMM Annual Conference Proceedings, AusIMM Darwin (1994), pp 109-115.
Back to top
Home
Lithium pegmatite fields without parental granites – an exemplary investigation (February 27, 2020)
Lithium is an alkali metal and an extremly lithophile element with a small atomic and ionic radius. With a density of 0.53 g/cm 3 it is the lightest metal of all (melting point at 180 0 C , boiling point 1336 0 C) but is too reactive for transport and use (PubChem 2020). Alloys with aluminium are employed in the aerospace industry. Chemical applications include lubricants (grease), pharmaceuticals, rechargeable and nonrechargeable lithium batteries, which have higher energy density and less weight compared to NiCd and NiMH. In the nuclear industry, 6Li is a starting material for the production of tritium, which is required for nuclear weapons and for thermonuclear fusion reactors. The latter are still in experimental stage but attractive because lithium fusion based on oceanic resources might provide humanity with energy for more than one million years. In nuclear fission power plants, lithium serves as a neutron absorber and heat exchanger.
It is commonplace knowledge that the electrification of vehicles, and large-scale electricity storage mainly rest on Li-ion batteries. Rapidly growing demand is forecast. Scientific research and prospecting activities profit from this situation.
Lithium concentrations of economic significance are restricted to few genetic classes (cited from W.L. Pohl 2020) :
- 1 Endogranitic magmatic-hydrothermal zinnwaldite-topaz greisen bodies in highly fractionated granite cupolas (e.g. Zinnwald, Germany);
- 2 Rare element (Li-Cs-Ta) pegmatites with lithium minerals and often, with exploitable tenors of Sn, Ta (>Nb) and Be (e.g. Greenbushes, W.A.); essentially, these mines sell concentrates of lithium minerals but Tanco (Canada), for example, is almost unique as a spodumene-Cs-Rb-Ta producer;
- 3 Evaporative lithium brines of playa lakes or from their subsurface aquifers (e.g. Salar de Atacama, Northern Chile; Salar de Uyuni, Bolivia); operations market lithium carbonate or chloride;
- 4 Solid lithium salt minerals (jadarite and zabuyelite) in terrestrial-lacustrine evaporites; lately discovered; a mine is in construction.
Lithium pegmatites are commonly related to parental granites sited in batholiths, although exceptions do occur: At the largest spodumene deposit of the world, the Greenbushes pegmatite in Western Australia, a parental granite remains elusive (Partington 2017). At the unique Tanco deposit (Canada) an petrogenetic link between a parent leucogranite and the pegmatite remains an assumption.
Before the profound investigations of Knoll et al. (2018), a similar situation without clear parental granites was discussed for the Permian Li-pegmatite province in the Eastern Alps (Central Europe). This metallogenetic belt extends over 400 km E-W in the Austroalpine basement of the Eastern Alps that was affected by the Carboniferous Variscan collision during the final welding of Pangea. The rocks and the outcrop shape of the lithium belt are controlled by a contractive deformation and high pressure amphibolite facies overprint during the Eoalpine collision and overthrusting in the Cretaceous (see Figure), when the Austroalpine acted as the upper plate.
During the Permian, the Adriatic ('African') plate was effected by lithospheric extension, basaltic underplating, high temperature-low pressure (HT/LP) metamorphism, of amphibolite to granulite facies, and by significant anatexis. The Li-pegmatites are one product of the latter.
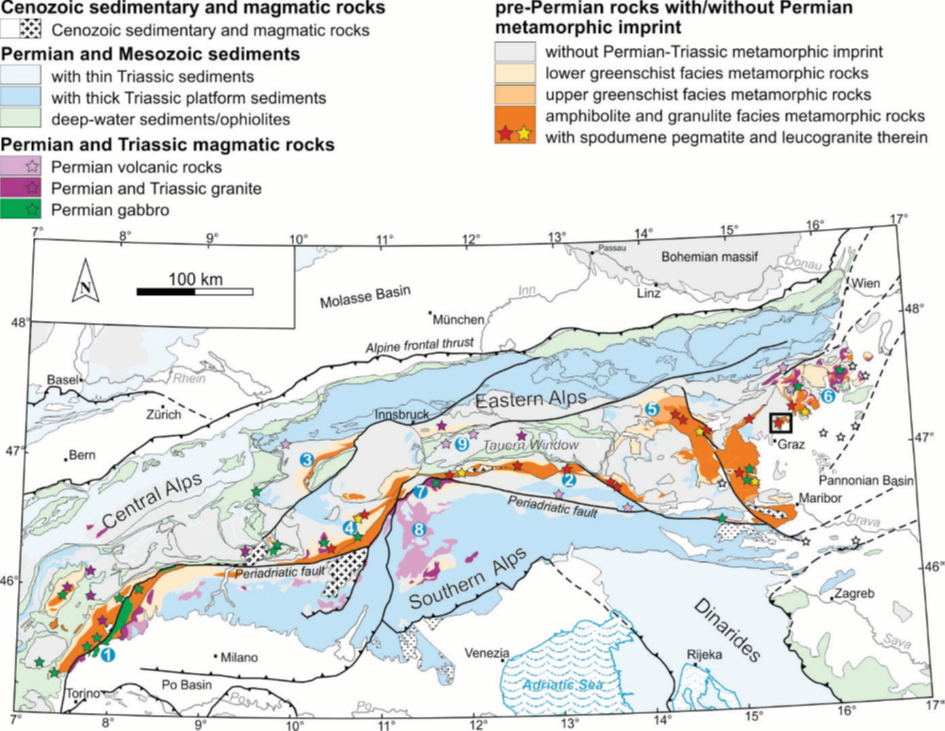
- Map of the Eastern Alps showing the distribution of Permian metamorphism and related magmatic and sedimentary rocks. Pegmatites occur in the amphibolite to granulite facies units. Black square indicates the EGU excursion area of Schuster et al. (2019). Credits to Schuster et al. (2019). Courtesy Tanja Knoll, GBA Vienna.
In the Austroalpine case, similar to Greenbushes, genetic relations between scattered small outcrops of leucogranites, common granitic, and Li-pegmatites remained doubtful. Resolving the doubts, Knoll et al. (2018) present a remarkably data-rich paper (based on field obervations, Rb-Sr and Sm-Nd geochronology, bulk-rock geochemistry and mineral Laser Ablation-ICPMS analyses). According to the authors, the results indicate a derivation of all magmatic rocks from anatectic melts generated during prograde HT/LP metamorphism of specific Al-rich metapelites, followed by fractionation into granitic pegmatite, leucogranite, moderately fractionated pegmatite and spodumene pegmatite. They show that trace elements of magmatic muscovites in leucogranites and mineralized pegmatites fall into a common fractionation trend, reaching tenors of moderately evolved pegmatites. Both leucogranites and Li-pegmatites are peraluminous. The latter display characteristics of the LCT family (rare-element class, albite-spodumene subtype), but certain doubts remain because characteristically, LCT pegmatites mark collision, supercontinent formation and the onset of cratonization (Bradley et al. 2017). In the petrotectonic classification or ore deposits, pegmatitic ore is part of the clan of Magmatogenic Ore Deposits (Pohl 2020).
The Austroalpine spodumene pegmatites occur in different shape and style: Dykes, boudins and pods are most common. Internal zoning is absent or faint. Source rocks are specific Al-rich metapelites. Host rocks are schists, paragneiss, marbles and amphibolites, the first locally displaying networks of felsic veinlets that indicate in situ anatectic melting. Migration of the melt seems to have been limited. In some outcrops, leucogranites, common granitic and Li-pegmatites occur closely together (Schuster et al. 2019), whereas globally, volatile-rich, highly fluid melts stable to relatively low temperatures migrate to great distances from their plutonic sources.
Possibly in the Austroalpine, melts of different character originated not only by differentiation and fractionation along migration but also by variable conditions of anatexis and selective mobilization of rare elements, such as availability of water and other volatiles, or partial melting at very low degrees (Robb 2005) pulsed by different temperatures. A supercritical and hydrous state of melts may have enhanced rare element enrichment; the investigation of melt inclusions in minerals may be used to reveal details of genetic processes (Thomas et al. 2019).
Of economic interest in the Permian East Alpine Li-pegmatite province is the Weinebene (Wolfsberg) spodumene deposit (Göd 1989) comprising several sizable dykes hosted in amphibolites and micaschists. ASX listed European Lithium is developing the deposit, based on a significant extension of resources compared to earlier work. Overall, the Permian Austroalpine lithium province remains underexplored.
References
Bradley, D.C., McCauley, A.D. & Stillings, L.M. (2017) Mineral-deposit model for lithium-cesium-tantalum pegmatites. U.S. Geological Survey Scientific Investigations Report 2010–5070–O, 48 pp.
European Lithium (2018) Wolfsberg Lithium Project. Accessed February 2020. URL https://europeanlithium.com/wolfsberg-lithium-project/project-geology-and-metallurgy/
Göd, R. (1989) The spodumene deposit at 'Weinebene', Koralpe, Austria. Mineralium Deposita 24, 270–278.
Knoll, T., Ertl, A., Giester, G., Horschinegg, M., Huet, B., Mali, H. & Onuk, P. (2018) Spodumene pegmatites and related leucogranites from the Austroalpine unit (Eastern Alps, Central Europe): Field relations, petrography, geochemistry, and geochronology. Canadian Mineralogist 56, 489-528. DOI: 10.3749/canmin.1700092
Partington, G.A. (2017) Greenbushes tin, lithium and tantalum deposit. Pp 153-157 in Australian ore deposits (ed G.N. Phillips), Australasian Institute of Mining and Metallurgy.
Pohl W.L. (in print, 2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. Schweizerbart Science Publishers, Stuttgart.
PubChem (2020) Periodic Table of Elements: Lithium. Accessed February 2020. URL https://pubchemdocs.ncbi.nlm.nih.gov/about
Robb, L.J. (2004) Introduction to Ore-Forming Processes. 384 pp Wiley Blackwell.
Schuster, R., Knoll, T., Mali, H., Huet, B. & Griesemeier, G.E.U. (2019) Field trip guide: A profile from migmatites to spodumene pegmatites (Styria, Austria). Berichte der Geologischen Bundesanstalt 134, 29 pp, Vienna.
Thomas, R., Davidson, P. & Appel, K. (2019) The enhanced element enrichment in the supercritical states of granite–pegmatite systems. Geochim. Cosmochim. Acta https://doi.org/10.1007/s11631-019-00319-z
Back to top
Home
Innovative Use of Triple Oxygen Isotope Geochemistry Illuminating the Genesis of Magnetite in Iron Oxide Apatite deposits. Open Access (January 16, 2020)
The application of isotope geochemistry provides ever more powerful tools for metallogenetic research. Isotope systems may indicate the source of water, gas, elements and various compounds in magmas and in hydrothermal solutions; they illuminate reactions with country rocks, expose mixing or differentiation processes, and they can reveal formation temperatures. Isotope geochemistry is an essential element of the quest for a full understanding of genetic processes.
The study by Peters et al. (2019) applies oxygen isotopes to answer one of the open questions concerning the origin of Kiruna type iron oxide apatite (IOA) deposits, which are an important source of iron ore globally. The Kiruna mine in northern Sweden is considered as the type-deposit of orthomagmatic iron ore formation related to felsic melts; under conditions of high aH2O and fO2 an immiscible FeOx-Ca-P liquid is in equilibrium with melt of felsic composition, as shown by the industrial Fe-smelting process.
In contrast to the purely orthomagmatic view, the participation of fluids in IOA genesis is often suggested; an unusual chemical composition of parental magmas and fluids may be due to assimilation of evaporitic, carbonatic or phosphatic upper crustal rocks, or of seawater, or of saline basinal brines. Accordingly, two end-member hypotheses compete for recognition: i) magmatic-hydrothermal replacement models, and ii) orthomagmatic (± fluid-assisted) interpretations. Along a 'mixing line', hybrid models appear likely.
Peters' et al. (2019) innovation concerns the first use of triple oxygen isotope analysis to the genesis of magnetite in IOA deposits. We are all aware of the use of stable oxygen isotopes 16 O and 18 O (and varying δ 18 O by mass-dependent fractionation) in many applications of geology. Using the rare 17 O has not yet reached the mainstream earth sciences but as this paper shows, it promises to provide new answers. Natural oxygen is composed of the three stable isotopes 16 O (99.757 ± 1.6%), 17 O (0.038 ± 0.1%), and 18 O (0.205 ± 1.4%) (PubChem 2020). Mass-independently fractionated 17 O can be used as a tracer to the source as demonstrated by Peters et al. (2019).
Kiruna magnetite ore is cogenetic with Paleoproterozoic igneous host rocks formed at an active continental margin during the Svecokarelian orogeny. A regional metallogenetic study (Martinsson et al. 2016) concludes that Kiruna type ores did form from an iron-rich magma, although generally displaying a hydrothermal overprint. The authors suggest that at 1.89 – 1.88 Ga tholeiitic magmas underwent liquid immiscibility during fractionation and interaction with crustal rocks, including meta-evaporites, generating the felsic igneous rocks and the segregated magnetite-apatite ore-forming melts.

- Gol-e-Gohar no. 3 open pit exposing the first ore (640 Mt @ 53 % Fe) in 2015. Credits to Narges Alibabaie and Bernd Lehmann.
Peters et al. (2019) sampled magnetite from a swarm of iron ore deposits in the Early Cambrian Yazd-Sirjan IOA province in central Iran. Host rocks of iron ore are felsic igneous rocks. Country rocks include coeval (Hormuz) evaporites and limestones; dolomite xenoliths in volcanic host rocks suggest assimilation of evaporites that display low δ 17 O (their Fig. 3). The cloud of 23 data points in triple oxygen space includes orthomagmatic characteristics and is elongated to clearly evaporite-influenced values. Variable mixing is indicated.
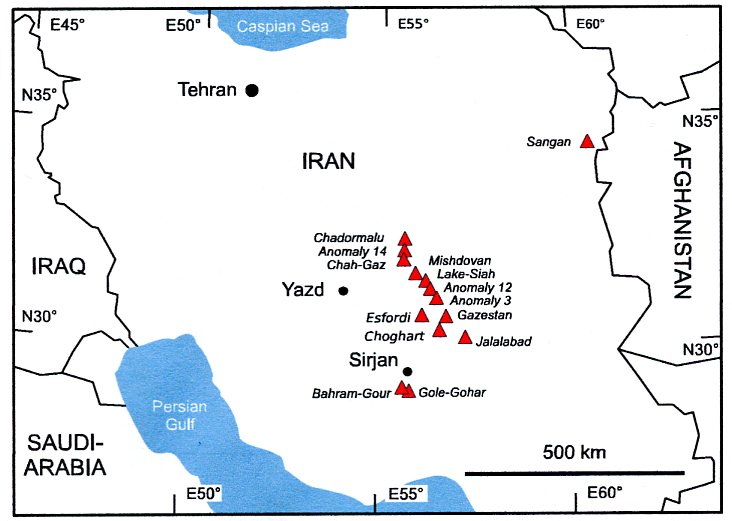
- Iron ore mines sampled in the Yazd-Sirjan Iron Oxide Apatite province in central Iran. Credits to Peters et al. (2019)
These findings seem to settle the discussion if evaporites may have a role in IOA deposit formation. Iron solubility in melts and brines is especially favoured. Generally, dissolution, assimilation and/or dehydration of salt-rich evaporites within basinal sediments are important agents facilitating ore formation (Pohl in print).
References
Martinsson, O., Billström, K., Broman, C., Weihed, P. & Wanhainen, Ch. (2016) Metallogeny of the Northern Norrbotten Ore Province, northern Fennoscandian Shield with emphasis on IOCG and apatite-iron ore deposits. Ore Geol. Rev. 78, 447 – 492.
Peters, S.T.M., Alibabaie, N., Pack, N., et al., Lehmann, B. (2020) Triple oxygen isotope variations in magnetite from iron-oxide deposits, central Iran, record magmatic fluid interaction with evaporite and carbonate host rocks. Geology 48, p. XXX – XXX, https://doi.org/10.1130/G46981.1 – Gold Open Access.
Pohl W.L. (in print, January 2020) Economic Geology, Principles and Practice: Metals, Minerals, Coal and Hydrocarbons – an Introduction to Formation and Sustainable Exploitation of Mineral Deposits. 2nd ed. Schweizerbart Science Publishers, Stuttgart.
PubChem (2020) National Center for Biotechnology Information. PubChem Database. URL https://pubchem.ncbi.nlm.nih.gov/element/Oxygen (accessed on Jan. 16, 2020).
Back to top
Home


